Health-&-Fitness
Urgency for vaccine grows as virus ravages China's pigs
Beijing, Aug 20 (AP/UNB) — Scientists are working to develop a vaccine to help guard the world's pork supply as a deadly virus ravages Asia's pig herds.
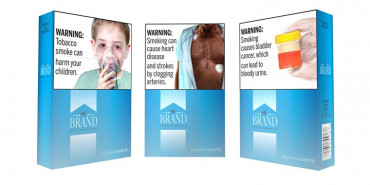
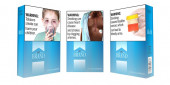
US makes new push for graphic warning labels on cigarettes
Washington, Aug 16 (AP/UNB) — U.S. health officials are making a new attempt at adding graphic images to cigarette packets to discourage Americans from lighting up. If successful, it would be the first change to U.S. cigarette warnings in 35 years.
FDA approves TB pill that cures more hard-to-treat patients
Trenton, Aug 15 (AP/UNB) — U.S. regulators Wednesday approved a new tuberculosis medicine that shortens and improves treatment for the hardest-to-treat cases, a worsening problem in many poor countries.
Foods to eat (and avoid) if you have dengue fever
Dhaka, Aug 9 (UNB) - Dengue fever is a mosquito-borne tropical disease caused by the dengue virus. Symptoms typically begin three to 14 days after the infection, and may include a high fever, headache, vomiting, muscle and joint pains, and a characteristic skin rash. When a mosquito bites a person infected with a dengue virus, the virus enters the mosquito. When the infected mosquito then bites another person, the virus enters that person’s bloodstream, reports The Indian Express.
Drinking three cups of coffee a day may increase migraine risk, says study
Dhaka, Aug 9 (UNB) - Drinking three or more cups of coffee daily may be associated with a higher risk of migraine, according to a study published Thursday, reports The Indian Express.
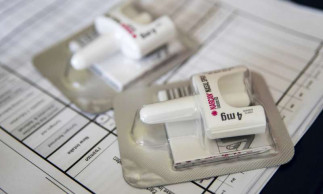
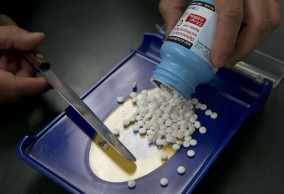
Boom in overdose-reversing drug is tied to fewer drug deaths
New York , Aug 7(AP/UNB) — Prescriptions of the overdose-reversing drug naloxone are soaring, and experts say that could be a reason overdose deaths have stopped rising for the first time in nearly three decades.
Uganda begins largest trial of experimental Ebola vaccine
Kampala, Aug 3 (AP/UNB) — Researchers in Uganda have launched the largest-ever trial of the experimental Ebola vaccine that is expected to be deployed in neighboring Congo, where a deadly outbreak has killed over 1,800 people.
US to set up plan allowing prescription meds from Canada
Washington, Aug 1 (AP/UNB) — The Trump administration said Wednesday it will create a way for Americans to legally and safely import lower-cost prescription drugs from Canada for the first time, reversing years of refusals by health authorities amid a public outcry over high prices for life-sustaining medications.
3 companies pay California $70 million for delaying drugs
Sacramento, Jul 30 (AP/UNB) — Three pharmaceutical companies collectively are agreeing to pay California nearly $70 million to settle allegations that they delayed drugs to keep prices high, California Attorney General Xavier Becerra said Monday.
Bedridden for 11 years until he cured himself
Dhaka, Jul 27 (UNB) - Doug Lindsay was 21 and starting his senior year at Rockhurst University, a Jesuit college in Kansas City, Missouri, when his world imploded.